India's economic engine is revving up, but navigating the global headwinds requires sharp strategy. The rupee's recent volatility against the dollar underscores the complexities facing Indian businesses, impacting everything from import-export trade to domestic inflation. This week alone, we've seen significant shifts in the IT sector, with major players announcing restructuring and a renewed focus on AI-driven solutions. Meanwhile, the burgeoning startup ecosystem continues to attract substantial funding, despite a cautious global investor sentiment. The government's recent policy announcements on infrastructure development and digitalization are poised to further shape the business landscape, creating both opportunities and challenges for entrepreneurs and established corporations alike. Understanding these market dynamics is crucial for success in today's rapidly evolving Indian economy. Stay informed with Abtak.com for the latest insights and analysis on key sectors including finance, manufacturing, and technology – empowering your business decisions in a dynamic market. Explore the complete business news section now to stay ahead of the curve.

New Delhi, Aug 12 (PTI) Drug firm Lupin on Tuesday said it has partnered with Switzerland-based Sandoz Group AG to commercialise its biosimilar ranibizumab across multiple regions. Under the terms of the agreement, Sandoz will oversee commercialisation of the product across the European Union (excluding Germany), Switzerland, Norway, Australia, Hong Kong, Vietnam, and Malaysia, the Mumbai-based drug maker said in a statement. Lupin will be responsible for manufacturing the product and for regulatory submissions, it added. As per the deal, Sandoz will hold exclusive marketing rights in most of the designated markets, except for France, Australia, Vietnam, and Malaysia, where it will have semi-exclusive marketing rights, Lupin Ltd said. Pursuant to another agreement executed between the two companies, Sandoz will acquire sole rights for commercialization of Lupin's biosimilar ranibizumab in Canada, while Lupin will manage its manufacture and regulatory filings, it added. Ranibizumab is a recombinant humanized IgG1 monoclonal antibody fragment that binds to and inhibits vascular endothelial growth factor A (VEGF-A). Its indications encompass the treatment of patients with neovascular (Wet) age-related macular degeneration (AMD), macular edema following retinal vein occlusion (RVO), diabetic macular edema (DME) among others. "This partnership underscores our shared vision to expand global access to cutting-edge biologic therapies and improve outcomes for underserved patients," Lupin President EMEA and Emerging Markets Thierry Volle said. Shares of the company were trading marginally up at Rs 1,946.30 apiece on BSE. PTI MSS MSS ANU ANU

Hyderabad: The Chintan Shivir on the Pharmaceutical Sector, organized by the Pharmaceutical Export Promotion Council of India (PHARMEXCIL), with the support of the Ministry of Commerce & Industry, Government of India, concluded successfully today in Hyderabad. This event marked the continuation of the series of discussions, following successful editions in Chandigarh and Ahmedabad, and served as a platform for strengthening India's pharmaceutical export agenda. The inaugural session, chaired by Shri Rajesh Agrawal, IAS, Commerce Secretary, Department of Commerce and Industry, Ministry of Comm ...Read More >

Mumbai: Dr Reddy's Laboratories has entered into a collaboration with Reykjavik, Ireland-based biotech company Alvotech to co-develop, manufacture and commercialise a biosimilar version of blockbuster cancer drug Keytruda for the global markets. Keytruda (generic name is pembrolizumab), manufactured and marketed by Merck & Co, is the most successful medicine, recording worldwide sales of $29.5 billion in 2024. It is used to treat multiple forms of cancer. Under the terms of the agreement, both companies will be jointly responsible for developing and manufacturing the biosimilar candidate and ...Read More >
New Delhi: Commerce secretary Rajesh Agrawal Saturday asked the pharmaceutical industry to reduce its dependence on critical imported raw materials by meeting atleast 80-90% of domestic pharmaceutical requirements through indigenous production. Addressing an event in Hyderabad, he said while India exports to around 200 countries, there remains significant scope for expansion and resilience-building through a stronger market presence. Emphasising the need to navigate an increasingly uncertain and geopolitically volatile global environment, he said: "The importance of ensuring greater self-rel ...Read More >

Hyderabad: CuraTeQ Biologics, a wholly owned subsidiary of Aurobindo Pharma, on Monday said it has reported positive top-line Phase 3 results for BP11, its investigational biosimilar to Xolair, or omalizumab.CuraTeQ said the study met all primary endpoints in patients with chronic spontaneous urticaria receiving the 300 mg dose, demonstrating high comparability with the reference product. The company said it plans to use the results to support regulatory submissions for chronic spontaneous urtica ...Read More >

New Delhi, Sep 26 (PTI) Indian Pharmaceutical Alliance (IPA) on Friday said US President Donald Trump's move to impose 100 per cent import tariffs on pharmaceutical drugs from October 1 will impact only patented and branded products, not generic medicines. Reacting to the development, IPA Secretary General Sudarshan Jain said in a statement, "It is not applicable to generics medicines." Trump's statement refers to patented and branded products supplied to US, he added. In his post on social media platform Truth Social, Trump wrote, "Starting October 1st, 2025, we will be imposing a 100% Tari ...Read More >

New Delhi: US President Donald Trump's decision to impose 100% tariffs on branded and patented drug exports has shaken Indian pharmaceutical markets, despite most of India's $10 billion US sales being in generics. The uncertainty lies in whether complex generics and specialty medicines will be included, a move that could directly impact leading players like Sun Pharma, Dr. Reddy's, Cipla, Zydus Lifesciences, and others. Stocks Facing Highest Exposure Dr. Reddy's emerges as the most vulnerable, with 47% of its earnings tied to the US market, projected at $1.5 billion in FY26. Sun Pharma follo ...Read More >

New Delhi, Jun 27 (PTI) Biocon Ltd on Friday said its arm Biocon Biologics has been granted a notice of compliance by Health Canada for its biosimilar Yesafili injection used to treat various ailments of the eye. "Health Canada has granted a Notice of Compliance (NOC) for Yesafili(aflibercept), a biosimilar to Eylea injection, in vial and prefilled syringe presentations, 2 mg/0.05 mL on June 26, 2025," a regulatory filing by Biocon said. This approval paves the way for the launch of Yesafili in Canada, scheduled for July 4, 2025, it said, adding Yesafili is the first biosimilar to Eylea to b ...Read More >

New Delhi, Jul 3 (PTI) Biocon Ltd on Thursday said its arm Biocon Biologics Ltd has been granted marketing authorisation by the European Commission for Vevzuo and Evfraxy biosimilars of Denosumab used in the treatment of different bone diseases. The marketing authorisation for the European Union (EU) follows a positive opinion issued by the European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP) on April 25, 2025, Biocon Ltd said in a regulatory filing. Vevzuo is authorised for the prevention of bone complications in adults with advanced cancer involving bone and th ...Read More >

Hyderabad, April 4 (RAHNUMA): The Government of India reaffirmed strong support for the pharmaceutical sector at a high-level Chintan Shivir organized by the Department of Commerce and Pharmexcil. The inaugural session was chaired by Rajesh Agrawal, Secretary, Department of Commerce. Senior officials including the Chairman of Pharmexcil and Joint Secretary from the Department of Pharmaceuticals addressed the gathering. The Commerce Secretary, in his address, highlighted the strong growth trajectory of India's pharmaceutical industry, noting that India, with nearly 18-19% of the world's popul ...Read More >

The Indian Pharmaceutical Alliance (IPA) has appointed Sharvil Patel of Zydus Lifesciences as its new President, with Glenn Saldanha of Glenmark as Vice President. This leadership transition occurs as the industry navigates GST reforms and strengthens its global position in specialty medicines. The new team aims to drive innovation, patient access, and quality healthcare solutions. The Indian Pharmaceutical Alliance (IPA) has appointed Sharvil Patel, Managing Director of Zydus Lifesciences as its new President. Patel succeeds Samir Mehta, Chairman, Torrent Group, the industry body said in an ...Read More >

Bhubaneswar: The Odisha government Tuesday unveiled Pharmaceutical and Medical Devices Policy-2025, aiming to attract investment of Rs 25,000 crore and create 1 lakh jobs by 2030. Chief Minister Mohan Charan Majhi launched the new policy here at the Odisha Pharma Summit-2025 in the presence of Industries Minister Sampad Chandra Swain and senior government officers. Speaking on the occasion, Industries Department's Additional Chief Secretary Hemant Sharma said India imports about 85 per cent of medical devices from other countries, and there is a huge opportunity for this sector. As a lot of ...Read More >

New Delhi: The government is working to ensure minimal disruption to supply chains amid global uncertainties, while remaining prepared for a potential dip in exports and imports, Commerce Secretary Rajesh Agrawal said on Saturday. Addressing the press conference on the sidelines of a 'Chintan Shivir' session in Hyderabad, Agrawal told the media that the initiative aims to bring together government and industry stakeholders to identify challenges and chart a clear roadmap. The official also stated that India is focusing on sustaining the growth trajectory of pharmaceutical exports. He said t ...Read More >
HYDERABAD: India's pharmaceutical exports touched nearly USD 28.29 billion by February this financial year, marking a 5.6% growth compared to last year, a top official said at the Chintan Shivir-Scaling up Pharma Exports programme held in Hyderabad on Saturday. Organised by the Pharmaceutical Export Promotion Council of India (Pharmexcil) with support from the Union government, the event was chaired by commerce secretary Rajesh Agarwal, who said India's pharmaceutical industry, valued at around USD 60 billion with nearly 50% exports, has emerged as a global leader due to its scale, cost compe ...Read More >

Mumbai, Oct 31 (SocialNews.XYZ) Actress Deepika Padukone, who has been a strong advocate for mental health awareness, reacted to cricketer Jemimah Rodrigues' recent revelation about battling anxiety. The 'Om Shanti Om' actress, known for openly sharing her own struggles with depression in the past, praised Jemimah for her honesty and courage in speaking up, highlighting the importance of conversations around mental well-being in sports and beyond. Taking to her Instagram Stories, Deepika Padukone reshared a video of cricketer Jemimah Rodrigues, in which the athlete opened up about her struggl ...Read More >

New Delhi, Apr 4 (PTI) Commerce Secretary Rajesh Agrawal on Saturday asked the pharmaceutical industry to reduce its dependence on critical imported raw materials and diversify export supply chains to better navigate global uncertainties. Addressing an event in Hyderabad on the sector, he said while India exports to around 200 countries, there remains significant scope for expansion and resilience-building through a stronger market presence. Emphasising the need to navigate an increasingly uncertain and geopolitically volatile global environment, he stressed: "the importance of ensuring grea ...Read More >

First & only immuno-oncology drug approved by various regulatory authorities around the world Dr. Reddy's Laboratories has announced the launch of Toripalimab in India. Toripalimab is a New Biological Entity (NBE). It is the only immuno-oncology drug approved by various regulatory authorities around the world such as United States Food and Drug Administration (USFDA), European Medicines Agency (EMA), Medicines and Healthcare products Regulatory Agency (MHRA), and others for the treatment of adults with recurrent or metastatic nasopharyngeal carcinoma (RM-NPC). In 2023, Dr. Reddy's entered in ...Read More >
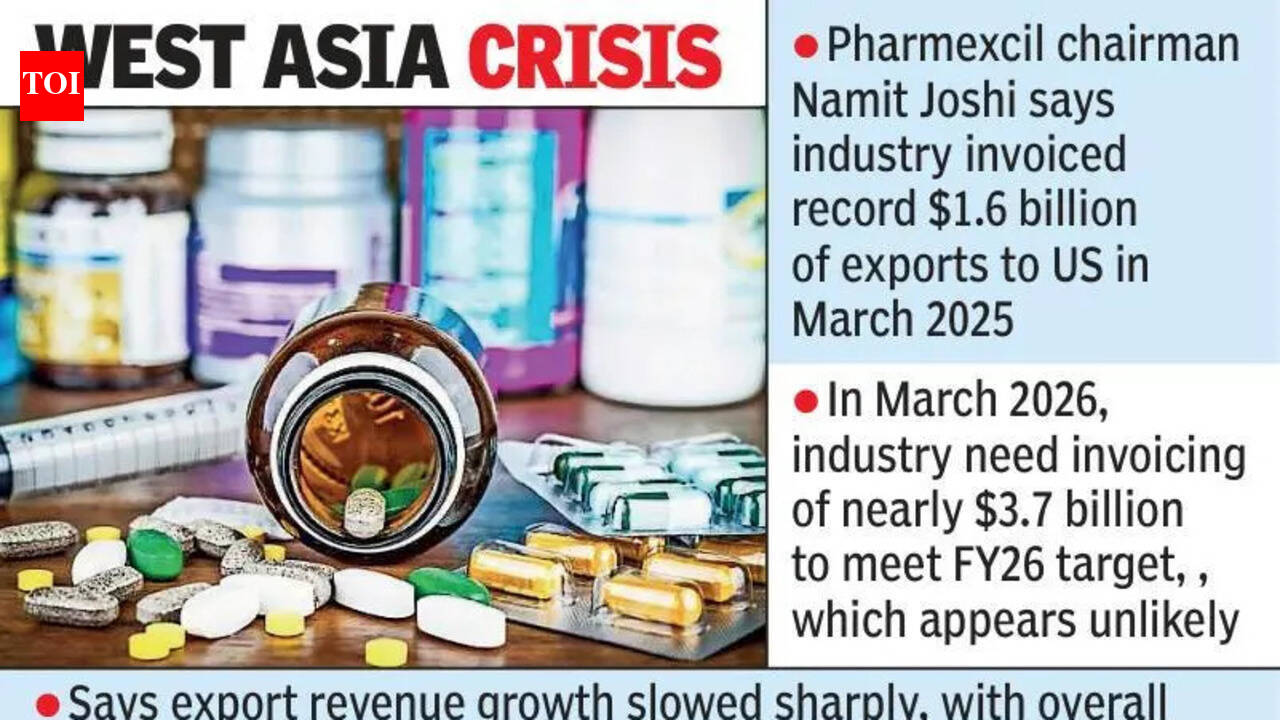
Hyderabad: India's pharma exports are likely to miss the FY26 target of $32 billion because of the West Asia crisis, though they should still exceed FY25 exports of $30.47 billion, Pharmexcil chairman Namit Joshi said at the Pharmexcil Chintan Shivir in Hyderabad.Joshi said the industry had invoiced a record $1.6 billion of exports to the US in March 2025, ahead of US tariffs that took effect in April 2025, compared with the usual monthly average of about $800 million. But in March 2026, the industry would need invoicing of nearly $3.7 billion to meet the FY26 target, which appears unlikely. H ...Read More >

Hyderabad, April 6 (RAHNUMA): CuraTeQ Biologics, a subsidiary of Aurobindo Pharma, has announced positive top-line results from its Phase 3 trial of BP11, an investigational biosimilar to Omalizumab (marketed as Xolair). The study met all primary endpoints, demonstrating strong comparability with the reference drug in treating chronic spontaneous urticaria (CSU) at a 300 mg dose. Conducted across 608 patients at nearly 80 sites in Europe and India, the trial showed precise equivalence in itch severity reduction at Week 12, aligning with requirements of both the Food and Drug Administration an ...Read More >

Dr. Mujahid Gilani (Senior Kashmiri Leader): Long before 14 August 1947, people of Jammu and Kashmir had voted in favour of Pakistan even before the division of Sub-Continent. They gave their decision over vehemently to become to join Pakistan as its Muslim majority province. In the backdrop of global situation, we must realize that people of Jammu and Kashmir are struggling for maintaining their lives. Their ratio of population in Jammu and Kashmir which was over 98 % in 1947 has been reduced and it is around 69%. The world has promised to Kashmiris to resolve Kashmir dispute through right to ...Read More >

New Delhi, Sep 26 (PTI) US President Donald Trump's move to impose 100 per cent import tariffs on pharmaceutical drugs from October 1 is unlikely to have an immediate impact on Indian exports, as it will affect only patented and branded products, not generic medicines, pharma industry players said on Friday. Indian Pharmaceutical Alliance (IPA), which represents 23 leading Indian pharmaceutical firms, including Dr Reddy's Laboratories, Sun Pharma, Lupin and Zydus Lifesciences, noted that Trump's latest tariff will not have implications for the generic drugmakers. In his post on social media ...Read More >

Striking a cautiously optimistic note, a senior official in the Commerce Ministry and representatives of the Pharmaceuticals Export Promotion Council of India on Saturday said FY26 pharma exports are expected to remain in the positive zone notwithstanding the West Asia war's impact on the March performance. For the 11 months upto February, the exports were nearly $28.29 billion as against the $26.79 billion in the corresponding period of previous fiscal. "We have already achieved 5.6% growth upt ...Read More >

New Delhi [India], October 31 (ANI): The U.S. Food and Drug Administration (FDA) has announced major steps to make it faster and less costly to develop biosimilar medicines, lower-cost "generic" alternatives to biologic drugs used to treat serious and chronic diseases. In a new draft guidance, the FDA has proposed major updates aimed at simplifying biosimilarity studies and reducing unnecessary clinical testing. The agency also launched a separate initiative to make it easier for biosimilars to be developed as interchangeable with brand-name biologics, allowing patients and pharmacists to ch ...Read More >

Chennai: The WTO's 14th Ministerial Conference (MC14) held in Cameroon ended without agreement on key issues, including e-commerce moratorium and expiry of safeguards against non-violation complaints against intellectual property policies. MC14 was one of the most inconclusive ministerial conferences in recent years. Despite WTO remaining ineffective in mitigating trade uncertainties, India to build coalitions to defend consensus-based decision-making at WTO, which is under threat due to growing plurilateral pacts, finds Ajay Srivastava, founder of Global Trade Research Initiative. Q) The WTO ...Read More >