Stay updated with the latest biosimilar news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on biosimilar topics.

Bengaluru: Biocon Limited has achieved a regulatory milestone in the United States, reinforcing its presence in the global diabetes treatment market. Biocon Pharma Limited, a subsidiary of Biocon Limited, has received approval from the U.S. Food and Drug Administration for Dapagliflozin Tablets in 5 mg and 10 mg strengths. The approval enables the company to market the drug for adults with type 2 diabetes mellitus, expanding its footprint in one of the world's largest pharmaceutical markets. T ...Read More >

New Delhi: Biocon Ltd on Tuesday announced the commercial launch of biosimilars Bosaya and Aukelso, indicated for different serious bone conditions, in the US. Bosaya (biosimilar to Prolia) and Aukelso (biosimilar to Xgeva) are now available by prescription nationwide through specialty pharmacies and healthcare providers, Biocon said in a statement. Also Read: Biocon not to rush generic semaglutide to packed India market, says incoming CEO Shreehas Tambe Both products have been previously app ...Read More >

New Delhi, Apr 7 (PTI) Biocon Ltd on Tuesday announced the commercial launch of biosimilars Bosaya and Aukelso, indicated for different serious bone conditions, in the US. Bosaya (biosimilar to Prolia) and Aukelso (biosimilar to Xgeva) are now available by prescription nationwide through specialty pharmacies and healthcare providers, Biocon said in a statement. Both products have been previously approved and granted interchangeable designation by the US Food and Drug Administration in Septembe ...Read More >

Hyderabad: CuraTeQ Biologics, a wholly owned subsidiary of Aurobindo Pharma, on Monday said it has reported positive top-line Phase 3 results for BP11, its investigational biosimilar to Xolair, or omalizumab.CuraTeQ said the study met all primary endpoints in patients with chronic spontaneous urticaria receiving the 300 mg dose, demonstrating high comparability with the reference product. The company said it plans to use the results to support regulatory submissions for chronic spontaneous urtic ...Read More >

Hyderabad, April 6 (RAHNUMA): CuraTeQ Biologics, a subsidiary of Aurobindo Pharma, has announced positive top-line results from its Phase 3 trial of BP11, an investigational biosimilar to Omalizumab (marketed as Xolair). The study met all primary endpoints, demonstrating strong comparability with the reference drug in treating chronic spontaneous urticaria (CSU) at a 300 mg dose. Conducted across 608 patients at nearly 80 sites in Europe and India, the trial showed precise equivalence in itch s ...Read More >

Hyderabad, April 4 (RAHNUMA): The Government of India reaffirmed strong support for the pharmaceutical sector at a high-level Chintan Shivir organized by the Department of Commerce and Pharmexcil. The inaugural session was chaired by Rajesh Agrawal, Secretary, Department of Commerce. Senior officials including the Chairman of Pharmexcil and Joint Secretary from the Department of Pharmaceuticals addressed the gathering. The Commerce Secretary, in his address, highlighted the strong growth traje ...Read More >

Two-year-old Bain Brandon has type 1 diabetes and needs insulin to live. But even with health insurance, the price tag isn't cheap. A 1-month supply of insulin vials and a 3-month supply of backup pens for the Mississippi toddler cost his parents $194 last week, according to his mom, 29-year-old Marlee Brandon. They can afford it right now -- but she worries about the future. "One day, Bain will be an adult, and he won't be able to be on our insurance anymore," she said. "I feel like a lot of ...Read More >

Hyderabad: The Chintan Shivir on the Pharmaceutical Sector, organized by the Pharmaceutical Export Promotion Council of India (PHARMEXCIL), with the support of the Ministry of Commerce & Industry, Government of India, concluded successfully today in Hyderabad. This event marked the continuation of the series of discussions, following successful editions in Chandigarh and Ahmedabad, and served as a platform for strengthening India's pharmaceutical export agenda. The inaugural session, chaired by ...Read More >
HYDERABAD: India's pharmaceutical exports touched nearly USD 28.29 billion by February this financial year, marking a 5.6% growth compared to last year, a top official said at the Chintan Shivir-Scaling up Pharma Exports programme held in Hyderabad on Saturday. Organised by the Pharmaceutical Export Promotion Council of India (Pharmexcil) with support from the Union government, the event was chaired by commerce secretary Rajesh Agarwal, who said India's pharmaceutical industry, valued at around ...Read More >
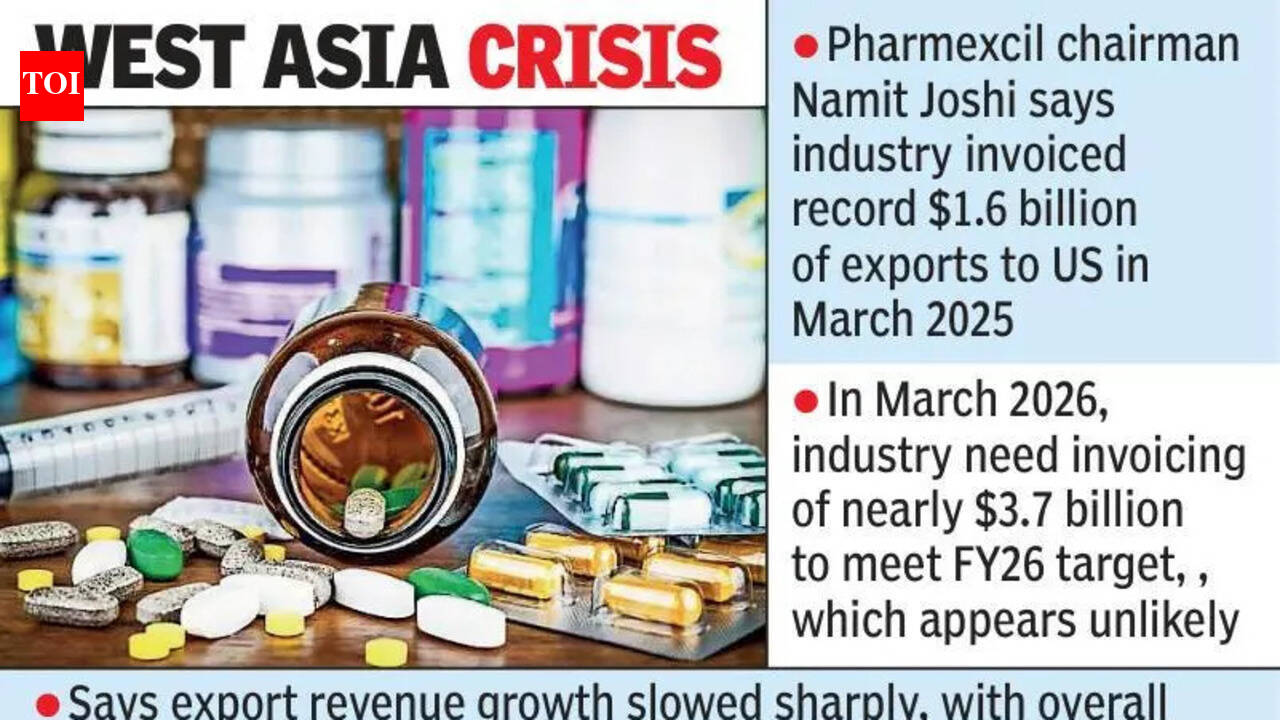
Hyderabad: India's pharma exports are likely to miss the FY26 target of $32 billion because of the West Asia crisis, though they should still exceed FY25 exports of $30.47 billion, Pharmexcil chairman Namit Joshi said at the Pharmexcil Chintan Shivir in Hyderabad.Joshi said the industry had invoiced a record $1.6 billion of exports to the US in March 2025, ahead of US tariffs that took effect in April 2025, compared with the usual monthly average of about $800 million. But in March 2026, the ind ...Read More >
New Delhi: Commerce secretary Rajesh Agrawal Saturday asked the pharmaceutical industry to reduce its dependence on critical imported raw materials by meeting atleast 80-90% of domestic pharmaceutical requirements through indigenous production. Addressing an event in Hyderabad, he said while India exports to around 200 countries, there remains significant scope for expansion and resilience-building through a stronger market presence. Emphasising the need to navigate an increasingly uncertain a ...Read More >

Striking a cautiously optimistic note, a senior official in the Commerce Ministry and representatives of the Pharmaceuticals Export Promotion Council of India on Saturday said FY26 pharma exports are expected to remain in the positive zone notwithstanding the West Asia war's impact on the March performance. For the 11 months upto February, the exports were nearly $28.29 billion as against the $26.79 billion in the corresponding period of previous fiscal. "We have already achieved 5.6% growth up ...Read More >

New Delhi: The government is working to ensure minimal disruption to supply chains amid global uncertainties, while remaining prepared for a potential dip in exports and imports, Commerce Secretary Rajesh Agrawal said on Saturday. Addressing the press conference on the sidelines of a 'Chintan Shivir' session in Hyderabad, Agrawal told the media that the initiative aims to bring together government and industry stakeholders to identify challenges and chart a clear roadmap. The official also sta ...Read More >

New Delhi, April 4 (SocialNews.XYZ) Union Minister Dr Jitendra Singh met with International Diabetes Federation President-elect Dr Niti Pall, and the latter highlighted that Asian and African countries "are increasingly looking to India for affordable diabetes care solutions," due to leadership in supplying cost-effective vaccines, an official statement said on Saturday. Dr Singh and Dr Pall discussed indigenous biosimilar insulin production and diabetes related indigenous biomanufacturing pros ...Read More >

New Delhi, Apr 4 (PTI) Commerce Secretary Rajesh Agrawal on Saturday asked the pharmaceutical industry to reduce its dependence on critical imported raw materials and diversify export supply chains to better navigate global uncertainties. Addressing an event in Hyderabad on the sector, he said while India exports to around 200 countries, there remains significant scope for expansion and resilience-building through a stronger market presence. Emphasising the need to navigate an increasingly unc ...Read More >

NEW DELHI, Apr 4 : "International Diabetes Federation" (IDF) President-elect, Dr Niti Pall, currently on India visit, called on Union Minister Dr Jitendra Singh, who is also a noted Professor of Medicine & Diabetes, and discussed, among other things, indigenous biosimilar Insulin production as well as Diabetes related indigenous biomanufacturing prospects. The meeting underscored the importance of strengthening India's capabilities in insulin manufacturing, particularly biosimilar insulins and ...Read More >

New Delhi [India], April 3 (ANI): The recent decision by US President Donald Trump to impose a 100 per cent tariff on patented pharmaceutical imports to US is unlikely to cause immediate disruption to Indian drugmakers, as the move largely targets branded medicines while exempting generics and biosimilars, said Siddharth Mittal, Managing Director and CEO (Designate) Syngene International Limited. Also Read | US Pilot Rescued After Fighter Jet Shot Down in Iran; Helicopter Also Hit During Search ...Read More >

New Delhi: The recent decision by US President Donald Trump to impose a 100 per cent tariff on patented pharmaceutical imports to US is unlikely to cause immediate disruption to Indian drugmakers, as the move largely targets branded medicines while exempting generics and biosimilars, said Siddharth Mittal, CEO and Managing Director of Biocon Limited. In an exclusive interaction with ANI, Mittal said the tariff action is aimed at boosting domestic manufacturing in the United States but does not ...Read More >

New Delhi [India], April 3 (ANI): The recent decision by US President Donald Trump to impose a 100 per cent tariff on patented pharmaceutical imports to US is unlikely to cause immediate disruption to Indian drugmakers, as the move largely targets branded medicines while exempting generics and biosimilars, said Siddharth Mittal, CEO and Managing Director of Biocon Limited. Also Read | Mysuru Cloth Bag Scam: Karnataka High Court Orders Probe Against IAS Officer Rohini Sindhuri Over INR 5.8 Crore ...Read More >

Bhubaneswar: The Odisha government Tuesday unveiled Pharmaceutical and Medical Devices Policy-2025, aiming to attract investment of Rs 25,000 crore and create 1 lakh jobs by 2030. Chief Minister Mohan Charan Majhi launched the new policy here at the Odisha Pharma Summit-2025 in the presence of Industries Minister Sampad Chandra Swain and senior government officers. Speaking on the occasion, Industries Department's Additional Chief Secretary Hemant Sharma said India imports about 85 per cent of ...Read More >

Mumbai, Oct 31 (SocialNews.XYZ) Actress Deepika Padukone, who has been a strong advocate for mental health awareness, reacted to cricketer Jemimah Rodrigues' recent revelation about battling anxiety. The 'Om Shanti Om' actress, known for openly sharing her own struggles with depression in the past, praised Jemimah for her honesty and courage in speaking up, highlighting the importance of conversations around mental well-being in sports and beyond. Taking to her Instagram Stories, Deepika Paduko ...Read More >

New Delhi [India], October 31 (ANI): The U.S. Food and Drug Administration (FDA) has announced major steps to make it faster and less costly to develop biosimilar medicines, lower-cost "generic" alternatives to biologic drugs used to treat serious and chronic diseases. In a new draft guidance, the FDA has proposed major updates aimed at simplifying biosimilarity studies and reducing unnecessary clinical testing. The agency also launched a separate initiative to make it easier for biosimilars t ...Read More >

The Indian Pharmaceutical Alliance (IPA) has appointed Sharvil Patel of Zydus Lifesciences as its new President, with Glenn Saldanha of Glenmark as Vice President. This leadership transition occurs as the industry navigates GST reforms and strengthens its global position in specialty medicines. The new team aims to drive innovation, patient access, and quality healthcare solutions. The Indian Pharmaceutical Alliance (IPA) has appointed Sharvil Patel, Managing Director of Zydus Lifesciences as i ...Read More >

New Delhi, Sep 26 (PTI) US President Donald Trump's move to impose 100 per cent import tariffs on pharmaceutical drugs from October 1 is unlikely to have an immediate impact on Indian exports, as it will affect only patented and branded products, not generic medicines, pharma industry players said on Friday. Indian Pharmaceutical Alliance (IPA), which represents 23 leading Indian pharmaceutical firms, including Dr Reddy's Laboratories, Sun Pharma, Lupin and Zydus Lifesciences, noted that Trump' ...Read More >

New Delhi: US President Donald Trump's decision to impose 100% tariffs on branded and patented drug exports has shaken Indian pharmaceutical markets, despite most of India's $10 billion US sales being in generics. The uncertainty lies in whether complex generics and specialty medicines will be included, a move that could directly impact leading players like Sun Pharma, Dr. Reddy's, Cipla, Zydus Lifesciences, and others. Stocks Facing Highest Exposure Dr. Reddy's emerges as the most vulnerable, ...Read More >

New Delhi [India], September 26 (ANI):Indian Pharmaceutical Alliance has said that the US President Donald Trump's announcement of tariffs of up to 100 per cent on imports of branded and patented pharmaceuticals starting October 1 specifically refers to patented or branded products, and not impact generic medicines. Indian Pharmaceutical Alliance (IPA) Secretary General Sudarshan Jain said, "The executive order refers to patented / branded products supplied to the US. It is not applicable to ge ...Read More >

New Delhi, Sep 26 (PTI) Indian Pharmaceutical Alliance (IPA) on Friday said US President Donald Trump's move to impose 100 per cent import tariffs on pharmaceutical drugs from October 1 will impact only patented and branded products, not generic medicines. Reacting to the development, IPA Secretary General Sudarshan Jain said in a statement, "It is not applicable to generics medicines." Trump's statement refers to patented and branded products supplied to US, he added. In his post on social me ...Read More >

New Delhi, Aug 12 (PTI) Drug firm Lupin on Tuesday said it has partnered with Switzerland-based Sandoz Group AG to commercialise its biosimilar ranibizumab across multiple regions. Under the terms of the agreement, Sandoz will oversee commercialisation of the product across the European Union (excluding Germany), Switzerland, Norway, Australia, Hong Kong, Vietnam, and Malaysia, the Mumbai-based drug maker said in a statement. Lupin will be responsible for manufacturing the product and for regu ...Read More >

New Delhi, Jul 16 (PTI) Biocon Biologics on Wednesday said it has received approval from the US health regulator to market a diabetes treatment medication. The company, a unit of Biocon Ltd, has received approval from the US Food and Drug Administration (USFDA) for Kirsty as the first and only interchangeable biosimilar to NovoLog (Insulin Aspart). Kirsty is a rapid-acting human insulin analog indicated to improve glycemic control in adults and pediatric patients with diabetes mellitus. The m ...Read More >

FDA Approves Kirsty (insulin aspart-xjhz), an Interchangeable Biosimilar to NovoLog BRIDGEWATER, N.J., United States and BENGALURU, Karnataka, India: July 15, 2025 -- Biocon Biologics Ltd (BBL), a fully integrated global biosimilars company and subsidiary of Biocon Ltd. (BSE code: 532523, NSE: BIOCON), today announced that the U.S. Food and Drug Administration (FDA) has approved Kirsty™ (Insulin Aspart-xjhz), 100 units/mL as the first and only interchangeable* biosimilar to NovoLog® (Insulin ...Read More >
Stay updated with the latest None news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on None topics.