Stay updated with the latest generic-drug news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on generic-drug topics.

New Delhi, Apr 9 (PTI) Glenmark Pharmaceuticals on Thursday said it has received final approval from the US health regulator for its generic version of progesterone vaginal inserts used in fertility treatments. The approval by the US Food and Drug Administration (USFDA) is for progesterone vaginal inserts of strength 100 mg, Glenmark Pharmaceuticals Ltd said in a regulatory filing. It has been determined by the USFDA to be bioequivalent and therapeutically equivalent to the reference listed dr ...Read More >

Ahmedabad: Zydus Lifesciences has expanded its presence in the U.S. generics market with a key regulatory approval for a widely used diabetes treatment. Zydus Lifesciences Limited has received final approval from the United States Food and Drug Administration for Dapagliflozin Tablets in 5 mg and 10 mg strengths. This approval enables the company to enter the U.S. market with a generic version of the drug, broadening its portfolio in a high-demand therapeutic category. With the approval, Zydus ...Read More >

New Delhi, Apr 8 (PTI) Zydus Lifesciences Ltd on Wednesday said it has received final approval from the US health regulator for its generic version of dapagliflozin tablets indicated to control blood sugar in adults with type 2 diabetes mellitus. The approval by the US Food and Drug Administration (USFDA) is for dapagliflozin tablets of strengths 5 mg and 10 mg, Zydus Lifesciences said in a regulatory filing. Dapagliflozin is indicated as an adjunct to diet and exercise to improve glycaemic co ...Read More >

New Delhi, Apr 7 (PTI) Aurobindo Pharma on Tuesday said it has received final approval from the US health regulator for its generic version of dapagliflozin tablets used to control blood sugar levels in people with type 2 diabetes. The final approval granted by the US Food & Drug Administration (USFDA) to manufacture and market dapagliflozin tablets is for strengths 5 mg and 10 mg, Aurobindo Pharma said in a regulatory filing. These are bioequivalent and therapeutically equivalent to the refer ...Read More >

Mumbai, Apr 6 (PTI) India's healthcare and pharmaceutical sector is set to enter a capability-led growth phase, potentially creating 2 to 2.5 million new jobs by 2030, while 30 to 35 per cent of the workforce will likely need reskilling, according to a report released on Monday. The country's healthcare and pharmaceutical sector is moving into a capability-led growth phase, with hiring increasingly focused on specialisation and technology integration across hospitals, AYUSH, home and elder care ...Read More >

Hyderabad, April 4 (RAHNUMA): The Government of India reaffirmed strong support for the pharmaceutical sector at a high-level Chintan Shivir organized by the Department of Commerce and Pharmexcil. The inaugural session was chaired by Rajesh Agrawal, Secretary, Department of Commerce. Senior officials including the Chairman of Pharmexcil and Joint Secretary from the Department of Pharmaceuticals addressed the gathering. The Commerce Secretary, in his address, highlighted the strong growth traje ...Read More >

Hyderabad: Aurobindo Pharma's board on Monday approved a Rs 800 crore share proposal to buy back up to 54.23 lakh fully paid-up equity shares of the company of face value Rs 1 each at Rs 1,475 a share.The proposed buyback, which is subject to regulatory and statutory approvals, represents up to 0.93% of the total number of equity shares in the company's total paid-up equity share capital.The Hyderabad-based generics drug maker informed the bourses that April 17, 2026, has been fixed as the recor ...Read More >
New Delhi [India], April 5 (ANI): US President Donald Trump has announced a sweeping 100 per cent tariff on select pharmaceutical imports, a move that comes shortly after a Jefferies report flagged the possibility of such measures, while maintaining a relatively constructive stance on Indian generic drug makers. In its April 2 report, Jefferies had noted that "the Trump administration is preparing to impose tariffs of 100 per cent on certain medicines," adding that the levies could be applied t ...Read More >
HYDERABAD: India's pharmaceutical exports touched nearly USD 28.29 billion by February this financial year, marking a 5.6% growth compared to last year, a top official said at the Chintan Shivir-Scaling up Pharma Exports programme held in Hyderabad on Saturday. Organised by the Pharmaceutical Export Promotion Council of India (Pharmexcil) with support from the Union government, the event was chaired by commerce secretary Rajesh Agarwal, who said India's pharmaceutical industry, valued at around ...Read More >
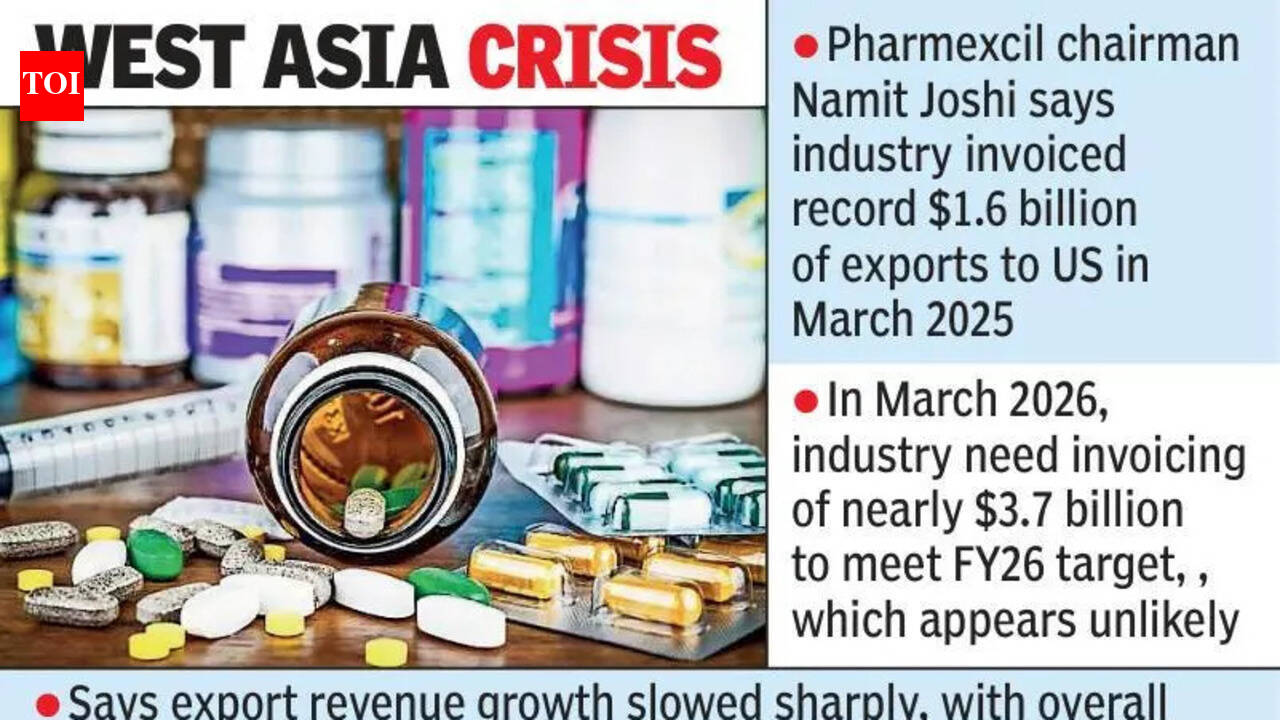
Hyderabad: India's pharma exports are likely to miss the FY26 target of $32 billion because of the West Asia crisis, though they should still exceed FY25 exports of $30.47 billion, Pharmexcil chairman Namit Joshi said at the Pharmexcil Chintan Shivir in Hyderabad.Joshi said the industry had invoiced a record $1.6 billion of exports to the US in March 2025, ahead of US tariffs that took effect in April 2025, compared with the usual monthly average of about $800 million. But in March 2026, the ind ...Read More >
New Delhi: Commerce secretary Rajesh Agrawal Saturday asked the pharmaceutical industry to reduce its dependence on critical imported raw materials by meeting atleast 80-90% of domestic pharmaceutical requirements through indigenous production. Addressing an event in Hyderabad, he said while India exports to around 200 countries, there remains significant scope for expansion and resilience-building through a stronger market presence. Emphasising the need to navigate an increasingly uncertain a ...Read More >

Striking a cautiously optimistic note, a senior official in the Commerce Ministry and representatives of the Pharmaceuticals Export Promotion Council of India on Saturday said FY26 pharma exports are expected to remain in the positive zone notwithstanding the West Asia war's impact on the March performance. For the 11 months upto February, the exports were nearly $28.29 billion as against the $26.79 billion in the corresponding period of previous fiscal. "We have already achieved 5.6% growth up ...Read More >

New Delhi, Apr 4 (PTI) Commerce Secretary Rajesh Agrawal on Saturday asked the pharmaceutical industry to reduce its dependence on critical imported raw materials and diversify export supply chains to better navigate global uncertainties. Addressing an event in Hyderabad on the sector, he said while India exports to around 200 countries, there remains significant scope for expansion and resilience-building through a stronger market presence. Emphasising the need to navigate an increasingly unc ...Read More >

March 31 (Reuters) - Novo Nordisk has again cut the prices of its blockbuster diabetes and weight-loss drugs Ozempic and Wegovy by up to 36% and 48% in India, to fend off competition from cheaper generics made by local drugmakers. India's market for diabetes and weight-loss drugs is set for a shake-up after the Danish drugmaker's patent on semaglutide, the active component in Ozempic and Wegovy, expired on March 20. At least half a dozen Indian drugmakers, including Dr Reddy's, Zydus an ...Read More >

New Delhi [India], April 3 (ANI): The recent decision by US President Donald Trump to impose a 100 per cent tariff on patented pharmaceutical imports to US is unlikely to cause immediate disruption to Indian drugmakers, as the move largely targets branded medicines while exempting generics and biosimilars, said Siddharth Mittal, Managing Director and CEO (Designate) Syngene International Limited. Also Read | US Pilot Rescued After Fighter Jet Shot Down in Iran; Helicopter Also Hit During Search ...Read More >

New Delhi: India is unlikely to be significantly impacted by US President Donald Trump's announcement of 100 per cent tariffs on certain patented drugs, as the country primarily exports low-cost generic medicines to America, think tank GTRI said on Friday. Trump signed an executive order on April 2, announcing the imposition of a 100 per cent ad valorem duty rate on the import of certain patented pharmaceuticals and associated pharmaceutical ingredients. The order, issued on April 2, 2026, bui ...Read More >
Chennai: The US has imposed 100 per cent duty on the import of patented pharmaceuticals and associated pharmaceutical ingredients. Generic drugs have been kept out of the duty for the time being. But, if pharma companies increasingly seek evergreening of patents, they will see fewer off-patented drugs in the pipeline. "I have determined that it is necessary and appropriate to impose a 100 percent ad valorem duty rate on the import of patented pharmaceuticals and associated pharmaceutical ingred ...Read More >

New Delhi, Apr 3 (PTI) India is unlikely to be significantly impacted by US President Donald Trump's announcement of 100 per cent tariffs on certain patented drugs, as the country primarily exports low-cost generic medicines to America, think tank GTRI said on Friday. Trump signed an executive order on April 2, announcing the imposition of a 100 per cent ad valorem duty rate on the import of certain patented pharmaceuticals and associated pharmaceutical ingredients. The order, issued on April ...Read More >

New Delhi: The recent decision by US President Donald Trump to impose a 100 per cent tariff on patented pharmaceutical imports to US is unlikely to cause immediate disruption to Indian drugmakers, as the move largely targets branded medicines while exempting generics and biosimilars, said Siddharth Mittal, CEO and Managing Director of Biocon Limited. In an exclusive interaction with ANI, Mittal said the tariff action is aimed at boosting domestic manufacturing in the United States but does not ...Read More >

New Delhi [India], April 3 (ANI): The recent decision by US President Donald Trump to impose a 100 per cent tariff on patented pharmaceutical imports to US is unlikely to cause immediate disruption to Indian drugmakers, as the move largely targets branded medicines while exempting generics and biosimilars, said Siddharth Mittal, CEO and Managing Director of Biocon Limited. Also Read | Mysuru Cloth Bag Scam: Karnataka High Court Orders Probe Against IAS Officer Rohini Sindhuri Over INR 5.8 Crore ...Read More >
New Delhi [India], April 3 (ANI): US President Donald Trump's decision to impose a 100 per cent tariff on patented pharmaceutical imports--citing national security and supply chain risks following a Section 232 probe--is expected to have a mixed impact on India. Crucially for India, the proclamation clarifies that generic drugs--a segment in which Indian firms dominate globally--are exempt "at this time," adding that such products "shall not be subject to Section 232 tariffs." For India, the i ...Read More >
New Delhi [India], April 3 (ANI): US President Donald Trump's decision to impose a 100 per cent tariff on patented pharmaceutical imports--citing national security and supply chain risks following a Section 232 probe--is expected to have a mixed impact on India. Crucially for India, the proclamation clarifies that generic drugs--a segment in which Indian firms dominate globally--are exempt "at this time," adding that such products "shall not be subject to Section 232 tariffs." Also Read | Delh ...Read More >
HYDERABAD: Soon-to-be brides and grooms seeking shortcuts to shed pounds before the big day have become the latest consumer target for weight-loss drugs in India. New Delhi wellness clinic Klarity Skin Clinic touts a "Mounjaro bride" package, while other clinics have woven weight-loss injections into "pre-wedding" transformation packages typically focused on skin treatments and hairstyle makeovers. Also Read: Weight-loss drug boom: Will cheaper Ozempic generics shake up India's fitness and nut ...Read More >

Lupin announced the completion of its acquisition of VISUfarma B.V. (VISUfarma), a leading European specialty pharmaceutical company focused on ophthalmology, from GHO Capital Partners LLP (GHO). The acquisition represents a significant milestone in Lupin's strategy to expand its specialty care portfolio and strengthen its presence in Europe. The integration of VISUfarma has significantly expanded Lupin's ophthalmology portfolio to over 60 branded products spanning dry eye, glaucoma, eyelid hyg ...Read More >

India's biotech ecosystem has also expanded significantly, now comprising over 2,500 startups, around 100 incubators, more than 600 research institutes, and over 200 accredited laboratories India's biotech sector is entering a new phase of global relevance, with the country's bioeconomy expanding from $10 billion in 2014 to $195 billion in 2026 and projected to touch nearly $300 billion by 2033, according to a report by Endiya Partners. The report, India's Biopharma Moment: Perspectives from D ...Read More >

New Delhi, March 31 (SocialNews.XYZ) India's bioeconomy is approaching the $200 billion mark, which is a significant milestone in its evolution with a strong policy push and growing innovation pipeline signalling the next phase of growth, according to a report released on Tuesday. The report by Endiya Partners said that the nation's bioeconomy has expanded sharply from around $10 billion in 2014 to over $195 billion in 2026, now contributing nearly 5 per cent to the GDP. India, long recognised ...Read More >

Mumbai/Naples: Lupin has taken a step forward in expanding its U.S. generics portfolio, securing regulatory progress for a key injectable product used in surgical care. Lupin has received tentative approval from the U.S. Food and Drug Administration for its Abbreviated New Drug Application for Sugammadex Injection. The approval covers two dosage strengths -- 200 mg/2 mL and 500 mg/5 mL -- both in single-dose vial formats. The company's product has been deemed bioequivalent to Merck's Bridion i ...Read More >

Ahmedabad -- Senores Pharmaceuticals Limited today announced that it has entered into a definitive agreement to acquire 100% equity stake in Apnar Pharma Private Limited ("Apnar Pharma"), along with the acquisition of five Abbreviated New Drug Applications (ANDAs), marking a significant milestone in Senores' growth strategy across regulated pharmaceutical markets. Apnar Pharma, incorporated on December 12, 2014, is a privately held pharmaceutical company engaged in the licensing, development, a ...Read More >

Mumbai, Oct 31 (SocialNews.XYZ) Actress Deepika Padukone, who has been a strong advocate for mental health awareness, reacted to cricketer Jemimah Rodrigues' recent revelation about battling anxiety. The 'Om Shanti Om' actress, known for openly sharing her own struggles with depression in the past, praised Jemimah for her honesty and courage in speaking up, highlighting the importance of conversations around mental well-being in sports and beyond. Taking to her Instagram Stories, Deepika Paduko ...Read More >

New Delhi [India], October 31 (ANI): The U.S. Food and Drug Administration (FDA) has announced major steps to make it faster and less costly to develop biosimilar medicines, lower-cost "generic" alternatives to biologic drugs used to treat serious and chronic diseases. In a new draft guidance, the FDA has proposed major updates aimed at simplifying biosimilarity studies and reducing unnecessary clinical testing. The agency also launched a separate initiative to make it easier for biosimilars t ...Read More >
Stay updated with the latest None news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on None topics.