India's economic engine is revving up, but navigating the global headwinds requires sharp strategy. The rupee's recent volatility against the dollar underscores the complexities facing Indian businesses, impacting everything from import-export trade to domestic inflation. This week alone, we've seen significant shifts in the IT sector, with major players announcing restructuring and a renewed focus on AI-driven solutions. Meanwhile, the burgeoning startup ecosystem continues to attract substantial funding, despite a cautious global investor sentiment. The government's recent policy announcements on infrastructure development and digitalization are poised to further shape the business landscape, creating both opportunities and challenges for entrepreneurs and established corporations alike. Understanding these market dynamics is crucial for success in today's rapidly evolving Indian economy. Stay informed with Abtak.com for the latest insights and analysis on key sectors including finance, manufacturing, and technology – empowering your business decisions in a dynamic market. Explore the complete business news section now to stay ahead of the curve.

Mumbai/Naples: Lupin has taken a step forward in expanding its U.S. generics portfolio, securing regulatory progress for a key injectable product used in surgical care. Lupin has received tentative approval from the U.S. Food and Drug Administration for its Abbreviated New Drug Application for Sugammadex Injection. The approval covers two dosage strengths -- 200 mg/2 mL and 500 mg/5 mL -- both in single-dose vial formats. The company's product has been deemed bioequivalent to Merck's Bridion injection. It is indicated for reversing neuromuscular blockade caused by rocuronium bromide and vecuronium bromide in patients undergoing surgery. This approval strengthens Lupin's injectable product pipeline in the U.S. market. The drug addresses both adult and pediatric patients aged 2 years and above, widening its potential clinical use across surgical settings. By advancing regulatory approvals in the U.S., Lupin continues to build its presence in complex generics and specialty pharmaceuticals. The development aligns with its broader focus on expanding high-value offerings across global markets. The tentative approval marks a key regulatory milestone as the company awaits final approval for commercial launch.
India's pharmaceutical industry, valued at $50 billion, is booming as a global leader in generic drugs. With significant exports to 200+ countries, India meets a substantial portion of global demand for affordable medicine. Challenges include regulatory hurdles and quality assurance, as the nation aims to expand its generic drug market. The Indian pharmaceutical industry, currently valued at $50 billion, is playing an increasingly crucial role on the global stage. Renowned as the 'pharmacy of the world,' India's pharma sector is a major exporter, supplying essential generic drugs to over 200 ...Read More >
HÀ NỘI While more than half of people living with rare diseases in Việt Nam are children, access to rare disease treatment remains limited due to high costs and a lack of comprehensive health insurance coverage. According to the Ministry of Health, Việt Nam has about six million people living with rare diseases, 58 per cent of whom are children. Data shows that an alarming 30 per cent of children with rare diseases die before the age of five. Rare diseases often include metabolic disord ...Read More >

New Delhi -- The World Health Organization (WHO) on Monday issued a health advisory warning against the use and distribution of three cough syrups believed to be responsible for the deaths of several children in India over the last month. The drugs were identified as specific batches of Coldrif, Respifresh TR and ReLife, the WHO said in a statement, manufactured by three Indian companies: Sresan Pharmaceutical, Rednex Pharmaceuticals, and Shape Pharma. The oral liquid drugs were found to conta ...Read More >
Hyderabad: Officials of the Telangana Drugs Control Administration (DCA), along with the Karkhana police, on Friday raided an unlicensed premises in Namalagundu of Secunderabad following a tip-off about the illegal sale of prescription drugs. During the raid, officials seized 66 vials of 'Termin' and 'Termiva' injections from the premises, which was being run by one M Naresh.According to officials, the injections were being stocked and sold illegally to gym-goers for bodybuilding and performance ...Read More >

Bengaluru: Biocon Limited has achieved a regulatory milestone in the United States, reinforcing its presence in the global diabetes treatment market. Biocon Pharma Limited, a subsidiary of Biocon Limited, has received approval from the U.S. Food and Drug Administration for Dapagliflozin Tablets in 5 mg and 10 mg strengths. The approval enables the company to market the drug for adults with type 2 diabetes mellitus, expanding its footprint in one of the world's largest pharmaceutical markets. The approved drug is indicated as an adjunct to diet and exercise to improve glycemic control in adul ...Read More >

New Delhi [India], October 8 (ANI): The Federation of All India Medical Association (FAIMA) has expressed serious concern over the reported deaths of children in Madhya Pradesh linked to the consumption of a particular cough syrup. In a letter to the Union Health Minister on Wednesday, the association stated that the incident raised serious questions about the safety, quality assurance, and monitoring mechanisms governing the manufacture and distribution of pharmaceutical products for children. FAIMA requested to constitute a Central Investigation Committee under the Ministry to conduct a de ...Read More >

The Enforcement Directorate (ED) on Monday launched searches at seven locations in Chennai in connection with the Coldrif cough syrup case, which led to the deaths of several children. The raids are being conducted under the provisions of the Prevention of Money Laundering Act (PMLA) and are linked to Sreesan Pharma, the manufacturer of the syrup that is now banned across several states in the country. Premises being searched by the central probe agency reportedly include residences of top offic ...Read More >

India's Antibiotic Habit: A Growing Threat to Gut HealthSource : AI-generated, for illustrative purposes Did you know? As per the University of Washington study published in The Lancet in 2021, drug-resistant infections in India were directly responsible for 267,000 deaths and contributed to nearly one million more. India stands at the cusp of an antibiotic epidemic that threatens the lives of thousands and poses a global health threat. There is a looming quick-fix culture of grabbing antibiotics, even for the common cold and fever. This habit is creating a hidden health crisis that undermin ...Read More >

Mumbai: Dr Reddy's Laboratories has entered into a collaboration with Reykjavik, Ireland-based biotech company Alvotech to co-develop, manufacture and commercialise a biosimilar version of blockbuster cancer drug Keytruda for the global markets. Keytruda (generic name is pembrolizumab), manufactured and marketed by Merck & Co, is the most successful medicine, recording worldwide sales of $29.5 billion in 2024. It is used to treat multiple forms of cancer. Under the terms of the agreement, both companies will be jointly responsible for developing and manufacturing the biosimilar candidate and ...Read More >

New Delhi: Chief minister Rekha Gupta on Sunday said authorities cannot go against demolition orders issued by courts and stressed that displaced residents have been provided accommodation.Her words came in the wake of the recent Madrasi Camp demolition for which the opposition Aam Aadmi Party (AAP) has criticised her. AAP has targeted Gupta, and alleged that the displaced Madrasi Camp residents were provided homes in faraway Narela, and those lack basic amenities.Attending an event to mark the construction of a Jan Sewa camp at a slum cluster in the Haiderpur area, Gupta told reporters: "In t ...Read More >

Srinagar- Congress MP Shashi Tharoor on Saturday said foreign countries were told clearly during the post-Operation Sindoor diplomatic outreach that in case of a fresh provocation, India would repeat its action. Speaking at a discussion at the Ahmedabad Management Association, Tharoor said Indian delegations which visited foreign countries in the aftermath of Operation Sindoor managed to convey that India acted with restraint and responsibility. "In other places, we actually were able to get some very high-placed people to say not only that they respected and supported India's right of self- ...Read More >

BusinessWire India Monrovia [Liberia]/ Mumbai (Maharashtra) [India], September 16: Merck Foundation, the philanthropic arm of Merck KGaA Germany, conducted the 7th Edition of Merck Foundation First Ladies Initiative - MFFLI Summit 2025 recently. It was inaugurated by Prof. Dr. Frank Stangenberg-Haverkamp, Chairman of Merck Foundation Board of Trustees, and Senator, Dr. Rasha Kelej, CEO of Merck Foundation & President of Merck Foundation First Ladies Initiative and H.E. Mrs. KARTUMU YARTA BOAKAI, First Lady of the Republic of Liberia and Ambassador of Merck Foundation "More Than a Mother" alon ...Read More >

New Delhi, Apr 3 (PTI) India is unlikely to be significantly impacted by US President Donald Trump's announcement of 100 per cent tariffs on certain patented drugs, as the country primarily exports low-cost generic medicines to America, think tank GTRI said on Friday. Trump signed an executive order on April 2, announcing the imposition of a 100 per cent ad valorem duty rate on the import of certain patented pharmaceuticals and associated pharmaceutical ingredients. The order, issued on April 2, 2026, builds on a Section 232 investigation launched on May 1, 2025, which cited national securit ...Read More >
New Delhi: Commerce secretary Rajesh Agrawal Saturday asked the pharmaceutical industry to reduce its dependence on critical imported raw materials by meeting atleast 80-90% of domestic pharmaceutical requirements through indigenous production. Addressing an event in Hyderabad, he said while India exports to around 200 countries, there remains significant scope for expansion and resilience-building through a stronger market presence. Emphasising the need to navigate an increasingly uncertain and geopolitically volatile global environment, he said: "The importance of ensuring greater self-rel ...Read More >

New Delhi, Sep 15 (SocialNews.XYZ) India's total exports of merchandise and services clocked a robust growth of 9.34 per cent in August to $69.16 billion compared to the same month of the previous year, according to official figures released by the Commerce and Industry Ministry on Monday. The country's merchandise exports during August 2025 went up to $35.10 billion as compared to $32.89 billion in August 2024 despite the economic uncertainties in the global market triggered by the US tariff hike. "Despite global uncertainties and trade policy uncertainties, Indian exporters have done well. ...Read More >

New Delhi, Sep 26 (PTI) Indian Pharmaceutical Alliance (IPA) on Friday said US President Donald Trump's move to impose 100 per cent import tariffs on pharmaceutical drugs from October 1 will impact only patented and branded products, not generic medicines. Reacting to the development, IPA Secretary General Sudarshan Jain said in a statement, "It is not applicable to generics medicines." Trump's statement refers to patented and branded products supplied to US, he added. In his post on social media platform Truth Social, Trump wrote, "Starting October 1st, 2025, we will be imposing a 100% Tari ...Read More >

New Delhi: US President Donald Trump's decision to impose 100% tariffs on branded and patented drug exports has shaken Indian pharmaceutical markets, despite most of India's $10 billion US sales being in generics. The uncertainty lies in whether complex generics and specialty medicines will be included, a move that could directly impact leading players like Sun Pharma, Dr. Reddy's, Cipla, Zydus Lifesciences, and others. Stocks Facing Highest Exposure Dr. Reddy's emerges as the most vulnerable, with 47% of its earnings tied to the US market, projected at $1.5 billion in FY26. Sun Pharma follo ...Read More >

Ministry of External Affairs (MEA) Official Spokesperson Randhir Jaiswal, reacting to US President Donald Trump's imposition of 100% tariffs on imports of branded and patented pharmaceutical drugs from October 1, 2025, said the government is monitoring the situation closely. On Thursday, the Trump administration announced that starting October 1, 2025, it will be imposing a 100% tariff on any branded or patented pharmaceutical product, unless a company is building their pharmaceutical manufacturing plant in America. Experts say Trump's decision is unlikely to have a significant impact on the ...Read More >

Mumbai, September 26: The Indian benchmark indices opened lower on Friday, following US President Donald Trump's announcement of tariffs on pharma imports and sustained selling by foreign institutional investors. As of 9.25 am, Sensex was down 388 points, or 0.48 per cent at 80,771 and Nifty was down 119 points, or 0.48 per cent at 24,771. Shares of Indian and other Asian pharmaceutical companies fell after Trump announced tariffs of up to 100 per cent on imports of branded and patented pharmaceutical drugs, starting October 1, 2025. Apart from drugs, President Trump announced a 50 per cent d ...Read More >

New Delhi [India], September 26 (ANI): India's pharmaceutical exports are not expected to face a major impact following the recent announcement of a 100 per cent tariff on branded and patented pharmaceutical products by the US, said Sudarshan Jain, General Secretary of the Indian Pharmaceutical Alliance (IPA). Speaking to ANI on Friday, Jain said that India's exports to the US primarily consist of generic drugs and active pharmaceutical ingredients (APIs), which are not covered under this tariff. "This [100 per cent tariff on imported pharmaceutical drugs] is applicable to patented branded p ...Read More >

For the month, FII sold equities worth Rs 30,141.68 crore, while DII bought equities worth Rs 55,736.09 crore. The broader indices broke a three-week gaining streak and underperformed the main indices as markets remained under pressure throughout the week on worries over fresh tariffs on pharma, higher visa fees by the US, persistent trade concerns, and continued FII selling. For the week, Nifty50 plunged 672.35 points or 2.65 percent to finish at 24,654.70, while the BSE Sensex index declined ...Read More >

New Delhi [India], April 3 (ANI): The recent decision by US President Donald Trump to impose a 100 per cent tariff on patented pharmaceutical imports to US is unlikely to cause immediate disruption to Indian drugmakers, as the move largely targets branded medicines while exempting generics and biosimilars, said Siddharth Mittal, Managing Director and CEO (Designate) Syngene International Limited. Also Read | US Pilot Rescued After Fighter Jet Shot Down in Iran; Helicopter Also Hit During Search. In an exclusive interaction with ANI, Mittal said the tariff action is aimed at boosting domestic ...Read More >
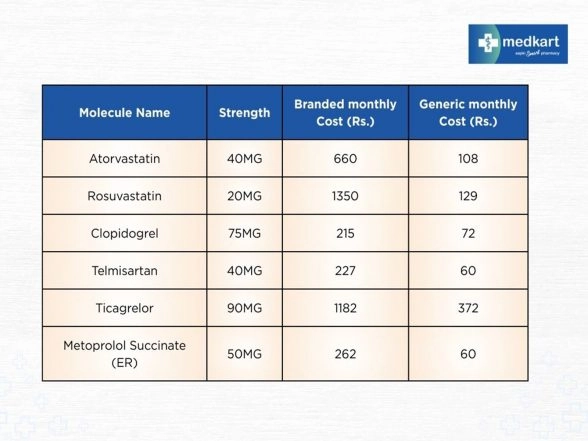
Ahmedabad (Gujarat) [India], September 29: As India observes World Heart Day, the conversation around cardiovascular health has once again turned to prevention, timely treatment, and most importantly, access to affordable medicines for everyone. Heart disease remains the leading cause of death in India at 28.1%, with more than half a billion people affected by a heart ailment. While treatment protocols have advanced, the cost of long-term medication continues to be a burden for many families. More so if one takes into account the fact that almost half of them cannot afford the medication. Als ...Read More >

The World Health Organization is seeking details from India regarding a cough syrup linked to child deaths. Several Indian states have banned the 'Coldrif' syrup. Lab reports confirmed it contained a toxic industrial solvent. A criminal case has been registered against the manufacturer, Sresan Pharma. The doctor who prescribed the syrup has been arrested. The World Health Organization on Wednesday sought clarification from Indian authorities on whether a cough syrup linked to deaths of children ...Read More >
New Delhi [India], April 3 (ANI): US President Donald Trump's decision to impose a 100 per cent tariff on patented pharmaceutical imports--citing national security and supply chain risks following a Section 232 probe--is expected to have a mixed impact on India. Crucially for India, the proclamation clarifies that generic drugs--a segment in which Indian firms dominate globally--are exempt "at this time," adding that such products "shall not be subject to Section 232 tariffs." Also Read | Delhi Police Files FIR Over Alleged FSSAI Document Tampering, Seeks X Account Details in Defamation Prob ...Read More >

New Delhi [India], April 3 (ANI): The recent decision by US President Donald Trump to impose a 100 per cent tariff on patented pharmaceutical imports to US is unlikely to cause immediate disruption to Indian drugmakers, as the move largely targets branded medicines while exempting generics and biosimilars, said Siddharth Mittal, CEO and Managing Director of Biocon Limited. Also Read | Mysuru Cloth Bag Scam: Karnataka High Court Orders Probe Against IAS Officer Rohini Sindhuri Over INR 5.8 Crore Irregularities. In an exclusive interaction with ANI, Mittal said the tariff action is aimed at bo ...Read More >
New Delhi, Dec 17 (PTI) Drug firm Granules India on Tuesday said its subsidiary has received approval from the US health regulator for a generic medication used to treat attention-deficit hyperactivity disorder. Granules Pharmaceuticals, Inc (GPI) has received approval from the US Food & Drug Administration (USFDA) for its abbreviated new drug application (ANDA) for Lisdexamfetamine Dimesylate chewable tablets, the drug firm said in a statement. The approved drug is available in multiple strengths; 10 mg, 20 mg, 30 mg, 40 mg, 50 mg, and 60 mg, it added. The company's product is the generic ...Read More >
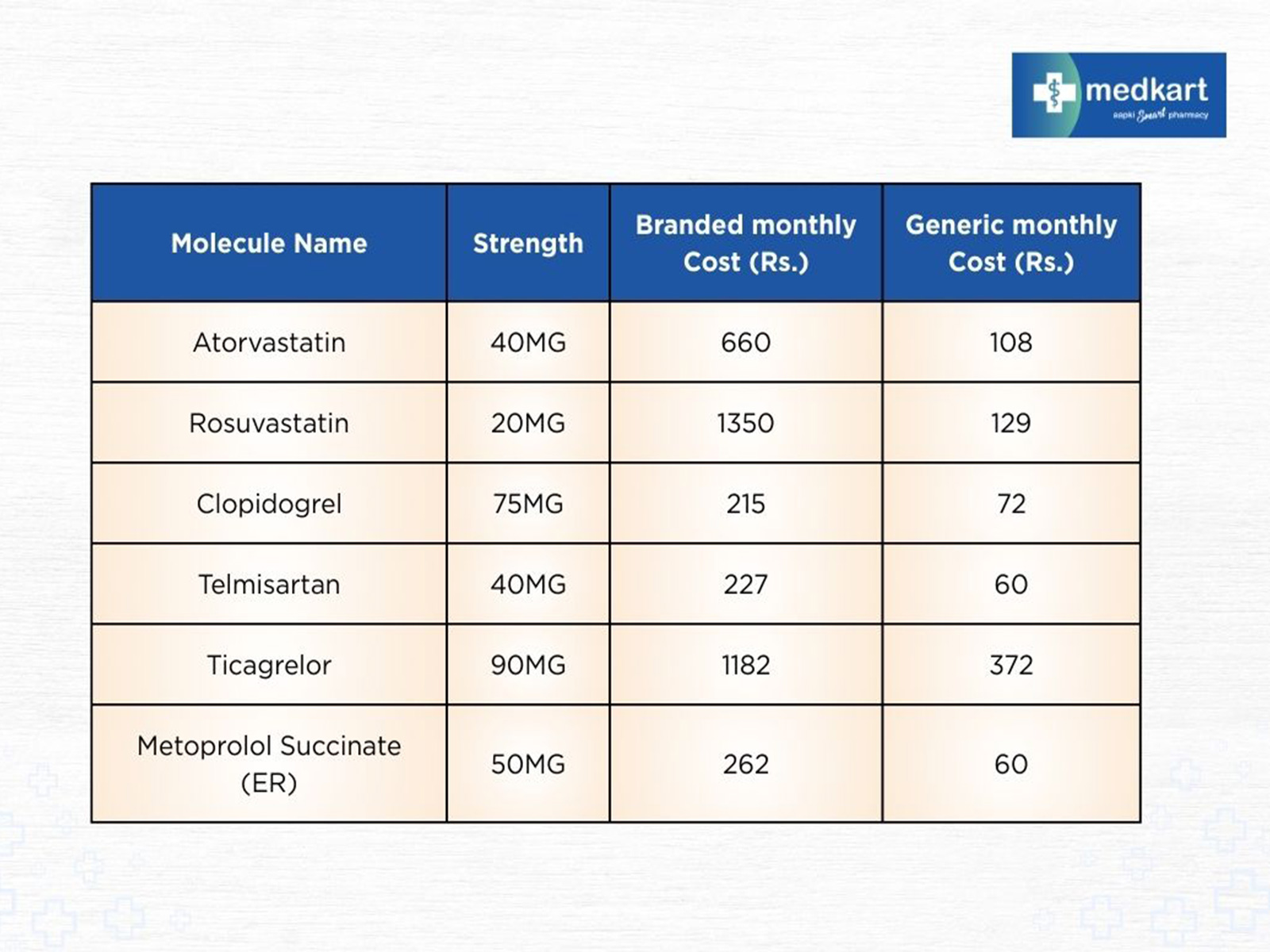
NewsVoir Ahmedabad (Gujarat) [India], September 29: As India observes World Heart Day, the conversation around cardiovascular health has once again turned to prevention, timely treatment, and most importantly, access to affordable medicines for everyone. Heart disease remains the leading cause of death in India at 28.1%, with more than half a billion people affected by a heart ailment. While treatment protocols have advanced, the cost of long-term medication continues to be a burden for many families. More so if one takes into account the fact that almost half of them cannot afford the medica ...Read More >

* Six children have died of kidney failure in Madhya Pradesh's Chhindwara district in 15 days * Investigators suspect contaminated cough syrup laced with toxic diethylene glycol as the cause of the deaths * Most of the victims had been administered Coldrif and Nextro-DS syrups, which have now been banned Did our AI summary help? Let us know. Switch To Beeps Mode In a heart-wrenching tragedy that has shaken Madhya Pradesh's Chhindwara district, six children have died of kidney failure in the ...Read More >

The Union health ministry addressed cough syrup quality. A meeting reviewed drug norm compliance. This followed child deaths linked to syrups in Chhindwara and Rajasthan. States received advice on cough syrup use for children. One cough syrup, Coldrif, contained DEG beyond limits. Action occurred against the manufacturer. Inspections began across 19 units. Officials urged adherence to manufacturing practices. Amid concerns over the quality of cough syrups, the Union health ministry on Sunday und ...Read More >