Stay updated with the latest diethylene-glycol news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on diethylene-glycol topics.

NEW DELHI: India's drug regulator has sought strict compliance with revised Schedule M. It has asked state drug regulators to carry out inspections of pharma manufacturing units which have applied for extension of Schedule M to verify their compliance with the requirements, in a bid to ensure quality of pharma products by mandating Good Manufacturing Practices (GMP). "In case any manufacturing unit is found non-complying to the requirements of revised Schedule M during inspections, strict actio ...Read More >

After contaminated cough syrups made headlines linked to child deaths in Madhya Pradesh and Rajasthan, India's drug regulator has launched a digital monitoring system so that officials can oversee production, movement and quality of high-risk ingredients used in making these formulations. The move comes after several reports of diethylene glycol (DEG) contamination in cough syrups, which raised serious safety concerns and prompted nationwide scrutiny of pharmaceutical manufacturing practices. ...Read More >
India is once again reeling from a preventable tragedy: the death of 25 children in Madhya Pradesh and Rajasthan due to contaminated cough syrup. This grim pattern of recurring medical negligence and weak drug regulation can be traced as far back as 1986, when 14 patients died in Mumbai's JJ Hospital after consuming glycerol contaminated with 18.5 per cent diethylene glycol (DEG). Then followed 11 child deaths in Bihar (1988), 33 in Gurgaon (1998), and 12 in Jammu & Kashmir (2020). Internationa ...Read More >

Given that deaths have now been reported across multiple states, this cannot remain a state-specific inquiry. It needs a coordinated, multi-state probe under the supervision of central agencies such as the CBI and the Union Health Ministry Over the past month, at least 20 children in Madhya Pradesh and Rajasthan have lost their lives after consuming contaminated cough syrup. In MP's Chhindwara district alone, where nine deaths occurred, families reported a distressing similarity. The children f ...Read More >

New Delhi -- The World Health Organization (WHO) on Monday issued a health advisory warning against the use and distribution of three cough syrups believed to be responsible for the deaths of several children in India over the last month. The drugs were identified as specific batches of Coldrif, Respifresh TR and ReLife, the WHO said in a statement, manufactured by three Indian companies: Sresan Pharmaceutical, Rednex Pharmaceuticals, and Shape Pharma. The oral liquid drugs were found to cont ...Read More >
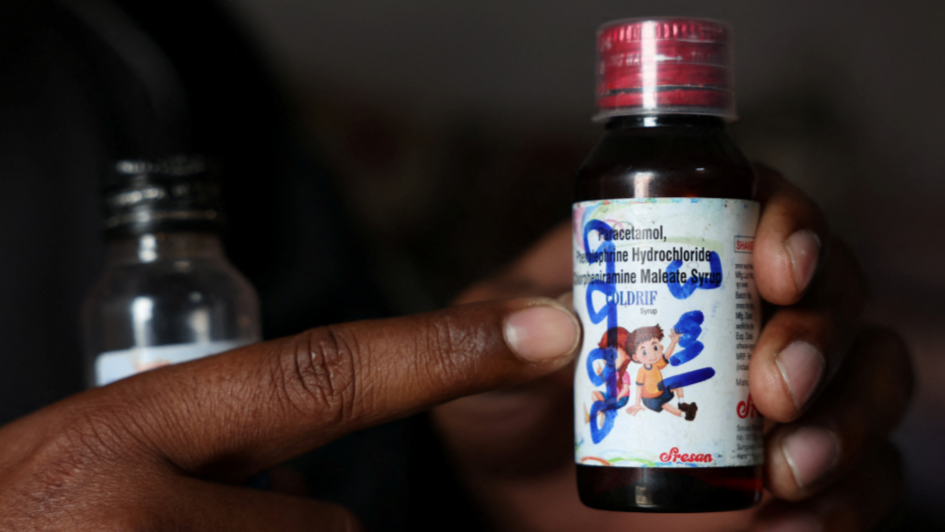
CHHINDWARA/NEW DELHI, India - India has refused drugmakers' requests to extend a year-end deadline for them to upgrade their manufacturing facilities to international standards, four sources said, amid public anger over the recent deaths of at least 24 children who consumed locally-produced cough syrup. New Delhi had in late 2023 ordered pharmaceutical companies to ensure their plants meet World Health Organization-recommended standards, which would require them to invest in protocols to preven ...Read More >

CHHINDWARA/NEW DELHI - India has refused drugmakers' requests to extend a year-end deadline for them to upgrade their manufacturing facilities to international standards, four sources said, amid public anger over the recent deaths of at least 24 children who consumed locally-produced cough syrup . New Delhi had in late 2023 ordered pharmaceutical companies to ensure their plants meet World Health Organisation-recommended standards, which would require them to invest in protocols to prevent cr ...Read More >

NEW DELHI: India has refused drugmakers' requests to extend a year-end deadline for them to upgrade their manufacturing facilities to international standards, four sources said, amid public anger over the recent deaths of at least 24 children who consumed locally-produced cough syrup. New Delhi had in late 2023 ordered pharmaceutical companies to ensure their plants meet World Health Organization-recommended standards, which would require them to invest in protocols to prevent cross-contaminati ...Read More >

Tragedy strikes Chhindwara as two more children die from adulterated cough syrup. The death toll now stands at 24. The victims, aged three years and nine months, succumbed to suspected renal failure in Nagpur. The WHO issued an alert against substandard cough syrups. Authorities have banned the formulation and arrested individuals involved. Manufacturing licenses have been revoked. The death toll in the adulterated cough syrup tragedy mounted to 24 after two more children from Chhindwara distri ...Read More >

Nagpur: The death toll in the toxic cough syrup, Coldrif, rose to 24 after the death of four-year-old girl, Ambika Vishwakarma, who battled for life for over a month at a private hospital in Nagpur early Wednesday.The Vishwakarma family, from Kakai Bhilwa village under Chourai tehsil in Chhindwara, lives around 60km southeast of Parasia block, a hotspot for Coldrif-related child deaths.The syrup, found to contain high levels of diethylene glycol (DEG), has led to multiple child fatalities. Two c ...Read More >

Bhopal/Nagpur: Doctors across the country have united to protest the arrest of paediatrician from Chhindwara, Dr Praveen Soni who is accused of prescribing the 'killer cough syrups' to kids resulting in their deaths.The Indian Academy of Paediatrics (IAP), representing over 47,000 paediatricians across the country on Friday sought withdrawal of cases against Dr Soni, claiming criminalising standard medical prescriptions will only create fear among doctors and severely impact healthcare delivery, ...Read More >

The Enforcement Directorate (ED) on Monday launched searches at seven locations in Chennai in connection with the Coldrif cough syrup case, which led to the deaths of several children. The raids are being conducted under the provisions of the Prevention of Money Laundering Act (PMLA) and are linked to Sreesan Pharma, the manufacturer of the syrup that is now banned across several states in the country. Premises being searched by the central probe agency reportedly include residences of top offi ...Read More >

CHENNAI: A week after reporting poor manufacturing and laboratory practices in Sresan Pharmaceuticals, the company that manufactured the lethal cough syrup that killed more than 20 children in Madhya Pradesh, the state health department suspended two former senior drug inspectors on charges of negligence for failing to inspect the unit, Health Minister Ma Subramanian said here on Thursday.Senior drug inspectors, Deepa Joseph and K Karthikeyan, who served in the Kancheepuram zone, were placed und ...Read More >

Tests revealed the presence of diethylene glycol -- a poisonous substance used in brake fluid and antifreeze -- known to cause acute kidney failure, especially in children. Follow TNM's channel for news updates and story links. Tamil Nadu Health Minister Ma Subramanian on Wednesday, October 8, said that the state government will initiate criminal action against the Kancheepuram-based pharmaceutical company that produced Coldrif, the contaminated cough syrup which caused the deaths of several c ...Read More >

The World Health Organization is seeking details from India regarding a cough syrup linked to child deaths. Several Indian states have banned the 'Coldrif' syrup. Lab reports confirmed it contained a toxic industrial solvent. A criminal case has been registered against the manufacturer, Sresan Pharma. The doctor who prescribed the syrup has been arrested. The World Health Organization on Wednesday sought clarification from Indian authorities on whether a cough syrup linked to deaths of children ...Read More >

Nagpur: In some cheer amidst the 45-day tragic trail left by the toxic cough syrup, one-year-old Pratik Pawar from Parasia block of Chhindwara, recovered from kidney failure and encephalopathy and returned home on Wednesday. Pawar, however, was administered a different brand of cough syrup, unlike others who consumed the toxic Coldrif that contained 48.6% diethylene glycol (DEG) -- a poisonous industrial chemical used in brake fluids and antifreeze.One more kid, Kunal Yaduwanshi, 3, from Chhindw ...Read More >

NEW DELHI, Oct 8 (Reuters) - Some Indian pharmaceutical firms failed to follow rules that every batch of medicinal ingredients is tested, the country's drug regulator said on Wednesday, opens new tab, after deaths in the past month of 17 children linked to toxic cough syrups. An advisory by the drugs controller general of India, Rajeev Raghuvanshi, said the regulator carried out checks at some factories and found serious lapses. In the advisory dated October 7 and posted on a government website ...Read More >

The affected children are from MP's Chhindwara, Rajasthan, and Maharashtra, while states like Kerala and Telangana have issued public alerts banning the use of Coldrif syrup. The Central Drugs Standard Control Organisation (CDSCO) has recommended the cancellation of the manufacturing licence of the Tamil Nadu-based pharmaceutical company that produced the cough syrup 'Coldrif', after tests confirmed the presence of diethylene glycol (DEG), a toxic substance, in the formulation, according to gov ...Read More >

Tirunelveli: Tamil Nadu Government will take criminal action against a drug manufacturer, whose cough syrup has been banned in the wake of children's deaths in Madhya Pradesh, Tamil Nadu minister Ma Subramanian said here on Wednesday. The government has issued its second notice against the drug manufacturer for the alleged presence of diethylene glycol in the cough syrup Coldrif, asking why criminal action should not be taken, the health and family welfare minister informed. The company has be ...Read More >

CHENNAI: The Tamil Nadu government has cancelled the manufacturing licences of Sresan Pharmaceuticals, the Kancheepuram-based firm that produced the toxic batch of Coldrif cough syrups linked to the deaths of 14 children in Madhya Pradesh. Of the children who died, 11 were from Parasia sub-division, two from Chhindwara city and one from Chaurai tehsil. On Sunday, the Parasia police arrested a government doctor for alleged negligence and registered a case against Sresan Pharmaceuticals, official ...Read More >

* Six children have died of kidney failure in Madhya Pradesh's Chhindwara district in 15 days * Investigators suspect contaminated cough syrup laced with toxic diethylene glycol as the cause of the deaths * Most of the victims had been administered Coldrif and Nextro-DS syrups, which have now been banned Did our AI summary help? Let us know. Switch To Beeps Mode In a heart-wrenching tragedy that has shaken Madhya Pradesh's Chhindwara district, six children have died of kidney failure in th ...Read More >

NEW DELHI: Following the tragic deaths of six children in Madhya Pradesh's Chhindwara district from acute kidney failure, the Union health ministry has launched a comprehensive, multi-agency investigation. A team from the National Centre for Disease Control (NCDC) has been deployed to the region, officials familiar with the matter said. Health officials strongly suspect a contaminated cough syrup is responsible for the fatalities. Preliminary reports suggest the over-the-counter medicine may ha ...Read More >
Stay updated with the latest None news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on None topics.