Stay updated with the latest central-drugs-standard-control-organisation news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on central-drugs-standard-control-organisation topics.

New Delhi, Apr 7 (PTI) As prices tumble and generic versions of GLP-1-based weight-loss drugs flood the market, a quiet frenzy is unfolding that has doctors and authorities alarmed. Medicines that are strictly prescription-based are being bought over the counter with their use expanding beyond clinical settings, often with little medical advice. For many people eager to lose weight quickly, the promise of fast results is overshadowing concerns about safety. Important factors -- like correct do ...Read More >

New Delhi [India], April 4 (ANI): The Indian Pharmacopoeia Commission ( IPC), which acts as the National Coordination Centre for the Pharmacovigilance Programme of India (PvPI) and provides evidence-based research to CDSCO, will be monitoring the safety of weight loss drugs. The Indian Pharmacopoeia Commission will be compiling a report of adverse events which are associated with the GLP-1 drugs. Also Read | Indian Railways Carries 1,670 MT Freight, Runs 25,000 Trains Daily in Financial Year 20 ...Read More >

New Delhi [India], April 4 (ANI): The Indian Pharmacopoeia Commission ( IPC), which acts as the National Coordination Centre for the Pharmacovigilance Programme of India (PvPI) and provides evidence-based research to CDSCO, will be monitoring the safety of weight loss drugs. The Indian Pharmacopoeia Commission will be compiling a report of adverse events which are associated with the GLP-1 drugs. According to the Official sources, the IPC has started the regular monitoring of adverse events of ...Read More >

NEW DELHI: India's drug regulator has sought strict compliance with revised Schedule M. It has asked state drug regulators to carry out inspections of pharma manufacturing units which have applied for extension of Schedule M to verify their compliance with the requirements, in a bid to ensure quality of pharma products by mandating Good Manufacturing Practices (GMP). "In case any manufacturing unit is found non-complying to the requirements of revised Schedule M during inspections, strict actio ...Read More >

After contaminated cough syrups made headlines linked to child deaths in Madhya Pradesh and Rajasthan, India's drug regulator has launched a digital monitoring system so that officials can oversee production, movement and quality of high-risk ingredients used in making these formulations. The move comes after several reports of diethylene glycol (DEG) contamination in cough syrups, which raised serious safety concerns and prompted nationwide scrutiny of pharmaceutical manufacturing practices. ...Read More >

New Delhi -- The World Health Organization (WHO) on Monday issued a health advisory warning against the use and distribution of three cough syrups believed to be responsible for the deaths of several children in India over the last month. The drugs were identified as specific batches of Coldrif, Respifresh TR and ReLife, the WHO said in a statement, manufactured by three Indian companies: Sresan Pharmaceutical, Rednex Pharmaceuticals, and Shape Pharma. The oral liquid drugs were found to cont ...Read More >
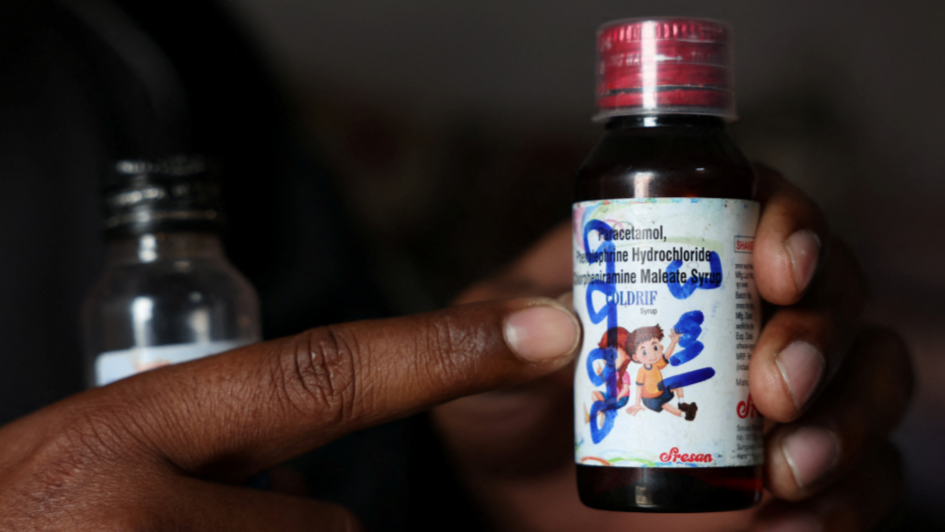
CHHINDWARA/NEW DELHI, India - India has refused drugmakers' requests to extend a year-end deadline for them to upgrade their manufacturing facilities to international standards, four sources said, amid public anger over the recent deaths of at least 24 children who consumed locally-produced cough syrup. New Delhi had in late 2023 ordered pharmaceutical companies to ensure their plants meet World Health Organization-recommended standards, which would require them to invest in protocols to preven ...Read More >

CHHINDWARA/NEW DELHI - India has refused drugmakers' requests to extend a year-end deadline for them to upgrade their manufacturing facilities to international standards, four sources said, amid public anger over the recent deaths of at least 24 children who consumed locally-produced cough syrup . New Delhi had in late 2023 ordered pharmaceutical companies to ensure their plants meet World Health Organisation-recommended standards, which would require them to invest in protocols to prevent cr ...Read More >

Amid growing demand for stricter compliance with safety and quality control norms for medical products, the Centre is framing a law to strengthen the legal framework for drug quality testing and market surveillance, along with regulation of medical devices and cosmetics. One of the major reasons behind the drafting of the law is the repeated complaints and concerns flagged by health regulators across the globe, including the WHO, over serious quality lapses by Indian drug manufacturers. The dr ...Read More >

The Enforcement Directorate (ED) on Monday launched searches at seven locations in Chennai in connection with the Coldrif cough syrup case, which led to the deaths of several children. The raids are being conducted under the provisions of the Prevention of Money Laundering Act (PMLA) and are linked to Sreesan Pharma, the manufacturer of the syrup that is now banned across several states in the country. Premises being searched by the central probe agency reportedly include residences of top offi ...Read More >

After the death of at least 20 children in Madhya Pradesh due to the consumption of contaminated cough syrup, the Central Drugs Standard Control Organisation (CDSCO) on Wednesday issued a strict advisory for drug controllers of all states and Union Territories and instructed them to ensure every batch of raw material and finished formulation is properly tested before use or sale. The advisory, dated October 7, 2025, was sent by Dr Rajeev Singh Raghuvanshi, Drug Controller General of India (DCGI ...Read More >

The affected children are from MP's Chhindwara, Rajasthan, and Maharashtra, while states like Kerala and Telangana have issued public alerts banning the use of Coldrif syrup. The Central Drugs Standard Control Organisation (CDSCO) has recommended the cancellation of the manufacturing licence of the Tamil Nadu-based pharmaceutical company that produced the cough syrup 'Coldrif', after tests confirmed the presence of diethylene glycol (DEG), a toxic substance, in the formulation, according to gov ...Read More >

New Delhi [India], October 8 (ANI): The Federation of All India Medical Association (FAIMA) has expressed serious concern over the reported deaths of children in Madhya Pradesh linked to the consumption of a particular cough syrup. In a letter to the Union Health Minister on Wednesday, the association stated that the incident raised serious questions about the safety, quality assurance, and monitoring mechanisms governing the manufacture and distribution of pharmaceutical products for children. ...Read More >

New Delhi [India], October 8 (ANI): The Federation of All India Medical Association (FAIMA) has expressed serious concern over the reported deaths of children in Madhya Pradesh linked to the consumption of a particular cough syrup. In a letter to the Union Health Minister on Wednesday, the association stated that the incident raised serious questions about the safety, quality assurance, and monitoring mechanisms governing the manufacture and distribution of pharmaceutical products for children. ...Read More >

New Delhi: Days after a pediatric doctor was arrested in Madhya Pradesh following the death of a few children after consumption of contaminated cough syrup, the Indian Medical Association (IMA) on Wednesday sought personal intervention of Health Minister JP Nadda over the issue and immediate withdrawal of the case against the bona fide medical practitioner. Meanwhile, the Central Drugs Standard Control Organisation (CDSCO) has asked all drug controllers from States and UTs for strict compliance ...Read More >

The Union health ministry addressed cough syrup quality. A meeting reviewed drug norm compliance. This followed child deaths linked to syrups in Chhindwara and Rajasthan. States received advice on cough syrup use for children. One cough syrup, Coldrif, contained DEG beyond limits. Action occurred against the manufacturer. Inspections began across 19 units. Officials urged adherence to manufacturing practices. Amid concerns over the quality of cough syrups, the Union health ministry on Sunday un ...Read More >

Chhindwara (MP), Oct 6 (PTI) The Madhya Pradesh police have formed a special investigation team (SIT) to probe the death of 14 children in Chhindwara due to suspected renal failure, linked to the consumption of a "toxic" cough syrup, officials said. Dr Praveen Soni from Chhindwara has been arrested for alleged negligence in connection with the child deaths, while a case has been registered against the Coldrif cough syrup manufacturing company, the officials said on Sunday. The body of the last ...Read More >

* Six children have died of kidney failure in Madhya Pradesh's Chhindwara district in 15 days * Investigators suspect contaminated cough syrup laced with toxic diethylene glycol as the cause of the deaths * Most of the victims had been administered Coldrif and Nextro-DS syrups, which have now been banned Did our AI summary help? Let us know. Switch To Beeps Mode In a heart-wrenching tragedy that has shaken Madhya Pradesh's Chhindwara district, six children have died of kidney failure in th ...Read More >

NEW DELHI: Centre on Friday issued an advisory to all states and Union territories calling for a "rational use of cough syrups in the paediatric population" after nine deaths were reported in Madhya Pradesh's Chhindwara due to kidney-related complications.The ministry of health and family welfare informed that the tests on the cough syrup samples revealed that it did not contain "Diethylene Glycol (DEG) or Ethylene Glycol (EG), contaminants that are known to cause serious kidney injury."Moreover ...Read More >

New Delhi, Sep 13 (SocialNews.XYZ) The Association of Indian Medical Device Industry (AiMeD) on Saturday welcomed the timely directions issued by the Department of Pharmaceuticals (DoP), National Pharmaceutical Pricing Authority (NPPA) and the Central Drugs Standard Control Organisation (CDSCO) to ensure smooth implementation of the revised GST rates on drugs, formulations, and medical devices, effective from September 22. The Office Memorandum issued by DoP-NPPA on Friday clarified that while ...Read More >

The District Magistrate of South Delhi has mandated CCTV installations in pharmacies to prevent illegal drug sales, focusing on Schedule H, H1, and X drugs. The All-India Organisation of Chemists and Druggists raises concerns over unregulated online pharmacies, urging swift action against illegal sales threatening public health. The District Magistrate of South Delhi has announced a new regulation mandating the installation of CCTV cameras in all pharmacies across the national capital. This ini ...Read More >

New Delhi: Apex drug regulatory body CDSCO has listed 17 drugs which should be flushed down the toilet rather than being thrown in dustbins if they have expired or are unused to help prevent harm to people and pets at home. These include tramadol, tapentadol, diazepam, oxycodone and fentanyl which may be especially harmful and in some cases fatal with just one dose if they are used by someone other than the person for whom the medicine was prescribed, the Central Drugs Standard Control Organisat ...Read More >

New Delhi, Jul 8 (PTI) Apex drug regulatory body CDSCO has listed 17 drugs which should be flushed down the toilet rather than being thrown in dustbins if they have expired or are unused to help prevent harm to people and pets at home. These include tramadol, tapentadol, diazepam, oxycodone and fentanyl which may be especially harmful and in some cases fatal with just one dose if they are used by someone other than the person for whom the medicine was prescribed, the Central Drugs Standard Cont ...Read More >

Mumbai, June 8 (SocialNews.XYZ) On her milestone 50th birthday, Shilpa Shetty took a heartfelt moment to reflect on her incredible journey and the defining stories that have shaped her both personally and professionally. Celebrating decades of success and growth, she shared insights into the experiences that made her the strong and inspiring woman she is today. Taking to Instagram, she posted a video of her transformation over the years and wrote, "Grateful for my journey and the stories that m ...Read More >

New Delhi [India], June 8 (ANI): In a major crackdown, the Anti-Narcotics Task Force (ANTF) of the Delhi Police has busted an interstate drug syndicate and seized 348.176 kilograms of Ganja, officials said. According to Apoorva Gupta, Deputy Commissioner of Police, ANTF, Crime Branch, Delhi, the seized contraband is valued at approximately Rs 1.75 crore in the international market. The drug was being smuggled from Andhra Pradesh to the Delhi-NCR region, concealed beneath a consignment of water ...Read More >

New Delhi [India], June 8 (ANI): In a major crackdown, the Anti-Narcotics Task Force (ANTF) of the Delhi Police has busted an interstate drug syndicate and seized 348.176 kilograms of Ganja, officials said. According to Apoorva Gupta, Deputy Commissioner of Police, ANTF, Crime Branch, Delhi, the seized contraband is valued at approximately Rs 1.75 crore in the international market. The drug was being smuggled from Andhra Pradesh to the Delhi-NCR region, concealed beneath a consignment of water ...Read More >

New Delhi [India], June 8 (ANI): In a major crackdown, the Anti-Narcotics Task Force (ANTF) of the Delhi Police has busted an interstate drug syndicate and seized 348.176 kilograms of Ganja, officials said. According to Apoorva Gupta, Deputy Commissioner of Police, ANTF, Crime Branch, Delhi, the seized contraband is valued at approximately Rs 1.75 crore in the international market. Also Read | Eknath Shinde Comes to the Rescue of Woman in Need of Kidney Transplant, Drops Her From Jalgaon to Mu ...Read More >
Stay updated with the latest None news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on None topics.