Stay updated with the latest essential-medicines news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on essential-medicines topics.
KURNOOL: Supplies of medicines in government hospitals across Andhra Pradesh are under severe strain, raising concerns over treatment for poor patients. Availability of essential drugs at state-run hospitals has dwindled significantly over the past few weeks due to rising input costs. Officials said global tensions, including the Iran war, have pushed up crude oil prices, increasing the cost of pharmaceutical inputs and disrupting supply. Drugs meant for the fourth quarter of 2025-26, expected ...Read More >
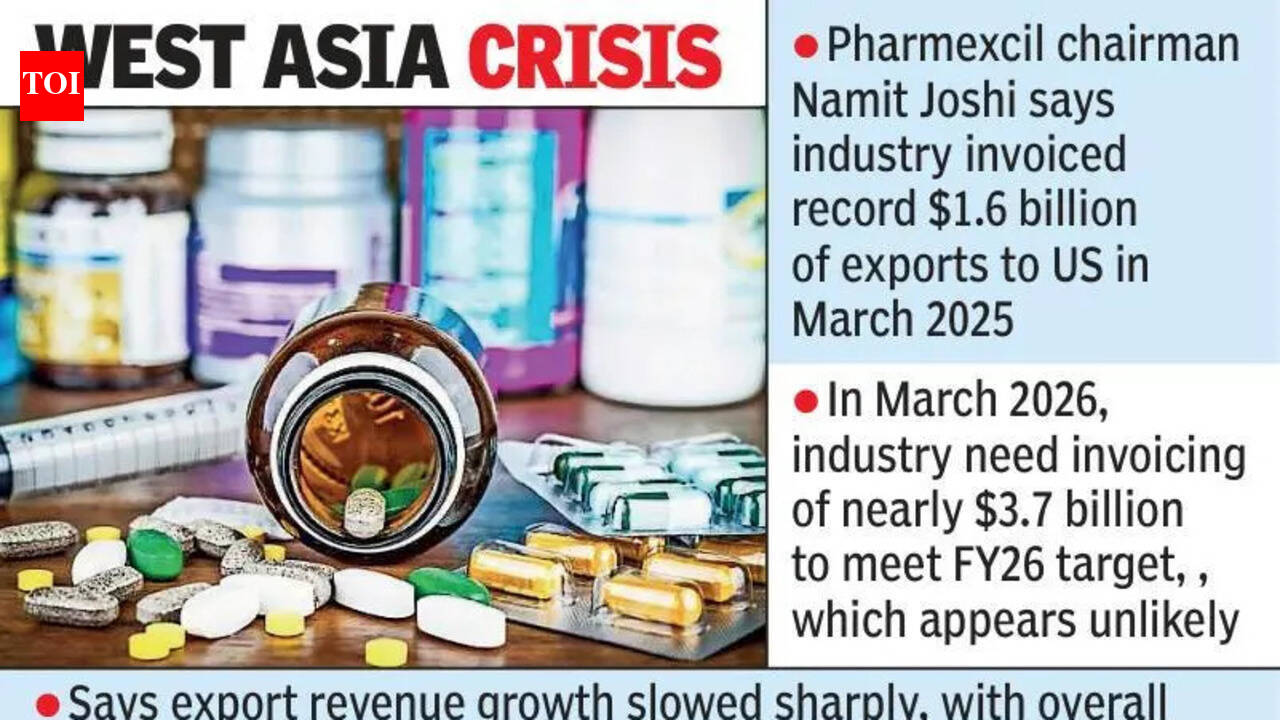
Hyderabad: India's pharma exports are likely to miss the FY26 target of $32 billion because of the West Asia crisis, though they should still exceed FY25 exports of $30.47 billion, Pharmexcil chairman Namit Joshi said at the Pharmexcil Chintan Shivir in Hyderabad.Joshi said the industry had invoiced a record $1.6 billion of exports to the US in March 2025, ahead of US tariffs that took effect in April 2025, compared with the usual monthly average of about $800 million. But in March 2026, the ind ...Read More >

New Delhi, Jul 3 (PTI) Biocon Ltd on Thursday said its arm Biocon Biologics Ltd has been granted marketing authorisation by the European Commission for Vevzuo and Evfraxy biosimilars of Denosumab used in the treatment of different bone diseases. The marketing authorisation for the European Union (EU) follows a positive opinion issued by the European Medicines Agency's Committee for Medicinal Products for Human Use (CHMP) on April 25, 2025, Biocon Ltd said in a regulatory filing. Vevzuo is auth ...Read More >
Stay updated with the latest None news, articles, and insights from trusted sources. Explore in-depth coverage and expert analysis on None topics.